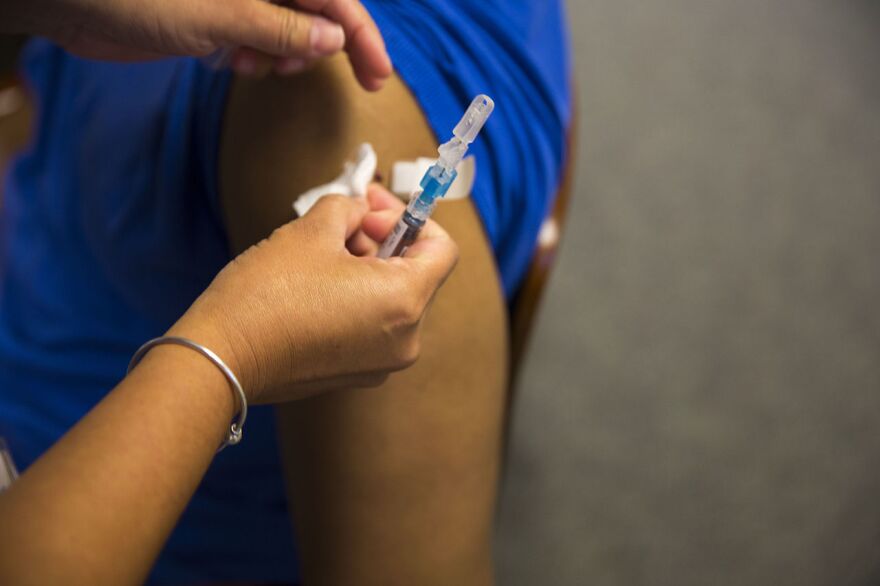
A new vaccine to protect against pneumonia and meningitis is being tested in people over 50. The Phase 3 trial of the experimental vaccine, made by the pharmaceutical giant Merck, is taking place internationally, with several sites in Florida.
The pneumococcal vaccine is typically recommended for older adults every five years. Other vaccines are on the market — including Pfizer's latest vaccine, Prevnar 20, which was approved by the FDA last year.
The current trial is testing how effective the Merck vaccine is against newer strains of the bacteria that have emerged around the world, mainly in Europe and Africa, said Dr. Jesus Navarro, who is leading one of the trial sites in Tampa.
"Those variants have really been reported in other countries, and they've caused a more severe disease, including death. So that's why this vaccine will be important," Navarro said.
He urged adults to stay up to date on their vaccines, including the COVID, flu and pneumococcal shots.
“Especially if they're immunocompromised," Navarro said. "Not to shy away from a vaccine that's every five years. And that will actually help them prevent mortality.”
Pneumococcal pneumonia causes some 150,000 hospitalizations each year, with symptoms like fever, coughing and chest pain.
Between 5% and 7% of cases result in death, a rate that “may be much higher among older adults or people with underlying medical conditions,” according to the Centers for Disease Control and Prevention.
About 2,000 people get pneumococcal meningitis each year, and the case fatality rate is 8% among children and 22% in adults, said the CDC.
Pneumococcal vaccines are needed because most people have the bacteria already in their mouths and can become sick when their immune systems are weakened, Navarro said.
"So what happens is, when you become immunocompromised, like elderly, children below 5, or people with HIV, diabetes, hypertension, kidney disease, then you become more susceptible to actually develop pneumonia, or meningitis," said Navarro.
People age 50 and above can take part in the Merck V116 trial, as long as they meet certain criteria, including never having received a pneumococcal vaccine before. Navarro said side effects include soreness at the injection site, and that the shot has been "generally well received from the patient standpoint."
More information can be found here and here, or call Genesis Clinical Research in Tampa at (813) 755-4403 or Clinical Research Trials of Florida at (813) 873-8102.
9(MDAyNDY5ODMwMDEyMjg3NjMzMTE1ZjE2MA001))
Copyright 2022 WUSF Public Media - WUSF 89.7